|
We were just notified that we had received funding for an NSF Biological Integration Institutes Design award. This award will allow me, Dawn Sumner, Patrick Shih, Titus Brown, David Britt, and Jonathan Eisen to spend 1.5 years designing an NSF Biological Integration Institutes Implementation proposal to understand the origin and evolution of photosynthesis. It will be a fantastic opportunity with a great group of folks!
0 Comments
I didn't plan to become a microbial ecologist. I stumbled into it when I took a graduate student field trip to the Rio Tinto as a masters student. I took the course, co-led by my eventual PhD advisor Jenn Macalady and Hiroshi Ohmoto, because I needed a course to fulfill a field work requirement, and it was the only one that fit my schedule. It was clear from the first minute that we stepped in the field that this was the work for me. I loved that every field site was a puzzle that required pieces from geochemistry, geology, and ecology to solve.
Five years later as I finished my PhD, I realized that no one had taken our field data and used it to make a paper. So I did, and the results were just published in the Geomicrobiology Journal. We describe the microbial community composition and geochemistry of Peña de Hierro, a pit lake in the Iberian Pyrite Belt and you can find it here. I am very grateful for this particular field course because it brought me to the field I love and taught me valuable field skills. I hope that in the future I will be able to provide my students similar opportunites that will give them the skills they need to succeed wherever their careers will take them. The wonderful David Lubarek wrote a feature about my work with geomicrobiologist Jenn Macalady and environmental engineer Bill Burgos. We worked to improve passive treatment of acid mine drainage in Pennsylvania waterways. Read the full article here.
This week marks the first week of school for students at UC Davis. Fellow Sumner lab member Marion Nachon and I are teaching a Freshman seminar on Astrobiology from an Earth (me) and Martian (Marion) perspective. We have an great group of students and I am really looking forward to the rest of the quarter.
For the first class I went over the Drake Equation. Instead of doing a traditional lecture, I did a modification of Philosophical Chairs and had the students make groups bases on their estimate for each value in the equation. Students would then discuss their reasoning amongst like-minded peers, and then explain to the other groups why they thought their value was correct. We would then regroup to so I could explain what value that research suggests (and why) and we would move on to the next part of the equation. It was really cool watching each group initially struggle with the question, cautiously suggest a reason to their small group, and then confidently tell the class why they thought their value was correct. I was also really impressed with their ability to reason through a question that they had never thought of before. For example, a large group of students hypothesized that intelligent life would only evolve on 10% of planets that already contained life. Initially individual students reasoning was "I don't know. It just seems unlikely" but by the end of their 3 minute discussion, they had decided that disturbances like meteor impacts, volcanic eruptions and climate change would make it difficult for life to evolve beyond "simple" life like bacteria. A (slightly pessimistic but) completely well reasoned argument (and from students who haven't taken any geology yet!). In case you were wondering our class chose the following values (though not everyone agreed) Known Values Stars formed per year = 7 (estimated from #stars in Milky Way/age of Milky Way) Stars with planets = 70% (estimated by Keplar mission) Planets per star = 1.6 Planets in the habitable zone = 24% (also from the Keplar mission) Unknown Values Fraction of habitable planets where life evolves = 10% (The class decided that the environmental conditions that are necessary for the origin of life are uncommon) Fraction of life containing planets that contain intelligent life = 10% (see reasoning above) We weren't able to fit in a discussion of the lifespan of a society or the fraction of intelligent societies that would engage in interstellar communication, but if we assume 10,000 years and 10% respectively , we come up with: ~19 societies in the Milky Way that are actively broadcasting. It looks like it is a lonely galaxy out there. The AGU GeoSpace blog featured my research with Dr. Trinity Hamilton and Dr. Jeff Havig (both of University of Minnesota). Check it out here.
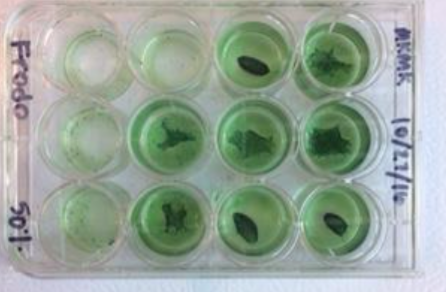
Our newest papers now available in JGR Biogeosciences. In the paper we present microbial communities in KY acid mine drainage and their ability to serve as analogs for microbial communities in the Neoarchean-Paleoproterozoic transition. The full text is free online here. You can see a few of our biofilms in the figure below.
Our paper on microbial communities and iron-oxidation rates at Scalp Level Run, PA came out in the most recent issue of Applied and Environmental Microbiology (link). Although AMD is a nasty environmental problem, I think that the sites can actually be quite beautiful. It looks like the editors at AEM agree and featured our AMD biofilms on the cover.
The Astrobiology Science Conference just posted the schedule for 2017 AbSciCon and I am very excited to be part of three great presentations.
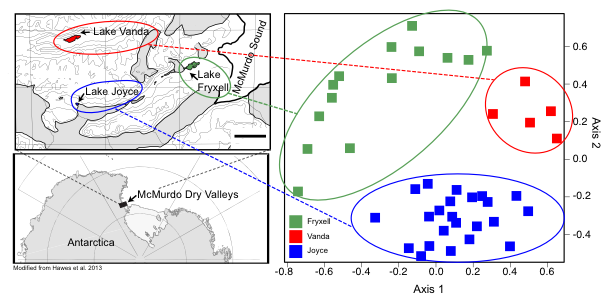
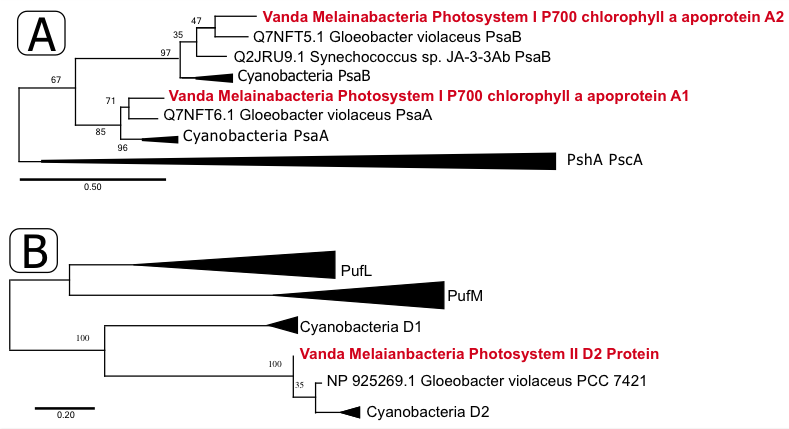
I am presenting my work on microbial communities in Antarctic lakes on Friday, April 28 (see the abstract here). My postdoc advisor, Dawn, is presenting some of our work on Melainabacteria and the evolution of oxygenic photosynthesis on Monday, April 24 (abstract; spoiler alert: it is super cool!) and Megan, an undergraduate researcher in our lab, is presenting her work at a poster session on Wednesday, April 26 (abstract). This morning, I got the great news that one of our papers was accepted at Applied and Environmental Microbiology and I could not be more exited. The preprint is available here. The paper focuses on a PA acid mine drainage (AMD) site called Scalp Level Run. We picked that site because it had the fastest iron-oxidation rate of any site we had studied before. We thought that it may contain some interesting iron-oxidizing microbes that could be used for AMD remediation. The microbial community at Scalp Level was dominated by two different OTUs (operational taxonomic units) from the genus Ferrovum. The only cultured Ferrovum sp. is an iron-oxidizing taxon so we assume that these two are also iron-oxiders. Interestingly, the two OTUs were inversely correlated, with one more abundant at the emergence of the AMD stream (where the concentration of iron is high) and the other at the base of the slope (where the concentration of iron is low). It seems as though the two taxa may be doing the same job while inhabiting different geochemical niche space. Cool! This paper was particularly fun because it included a great group of co-authors including my PhD advisor Jenn Macalady as well as Kyle Bibby (Pittsburgh) and Bill Burgos (Penn State), both of whom are environmental engineers. The paper also includes both with my unoffical mentor Dan (who helped me get up and running in my first year in the Macalady lab) and my undergraduate mentee Alex who did part of the research as her senior thesis.
It smells like a wet dog it here, but you can't blame me! If you haven't heard, we are getting a little bit of rain in California this week. Thankfully, Davis is not affected by flooding. Instead, we are minorly inconvenienced by the rain because biking, running, and walking in the downpour is, well, wet. Antecdotally, it seems like folks are driving a bit more and walking and biking a bit less to avoid the rain. Thus far, avoiding the outdoors hasn't been an option for me. My puppy LOVES the rain and turns in to the Tasmanian Devil if he doesn't spend enough time exercising outdoors. All that time playing in puddles means that everything smells like wet dog - my clothes, my car, my apartment, my couch - all of it. As it turns out, wet dog smell isn't actually from the dog per se. Instead, that stinky stench comes from microbial waste that is liberated by water. The American Chemical Society explains it all in their video below. If you don't want to hear other fun dog facts, fast foward to ~1:15. Find out why!
|








 RSS Feed
RSS Feed
